Bird migration
Which genes regulate this fascinating behaviour?
Among bird species, migration is the rule rather than the exception. Birds have the options and theoretical possibilities to choose various migratory strategies and make use of vastly diverse navigation and orientation systems. One characteristic of bird migration is its variability, both within and among species. Particularly fascinating are young birds on their first migratory journey covering thousands of kilometres that often spans continents. These tiny birds travel to wintering areas they have never been before - without the guidance of their parents, but with amazing accuracy! How do they do this?
Integrative approach
From quantitative genetics analysis of selection experiments we know that variation in migratory behaviour, as well as physiological and morphological adaptations to migration, are largely due to genetic differences. But so far we know nothing about the number and identity of genes involved in controlling migratory traits, or the magnitudes of their effects.
With our research we want to identify and map the genes behind the components shaping the migratory phenotype. We want to understand: What is the genetic basis of migration, and which signalling pathways are associated with variation of the migratory phenotype?
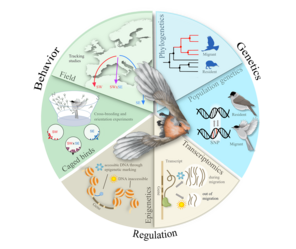
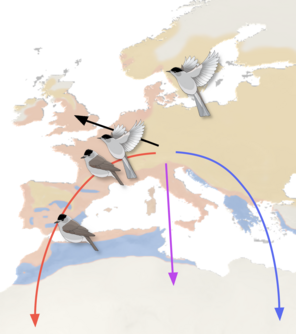
Our primary objective is to identify and characterise genetic variation and detect variation in gene expression patterns between blackcap populations following different migratory strategies, e.g. different migratory directions, long and short distance migration, and at least one completely sedentary population. To most efficiently address these questions, we combine high throughput sequencing techniques with state-of-the-art tracking technology, carefully designed behavioural experiments and solid knowledge of a well characterised study species.
Specifically, we identify and map the genes behind the components shaping the migratory phenotype using whoe genome sequencing approaches. Because the basis of phenotypic differences might not only lay in the sequence, we complement the sequencing approach with gene expression profiling and characterisation of chromatin modification to investigate the extent of phenotypic variation manifested by expression differences, either through slight genetic differences or regulatory epigenetic processes.
AG Liedvogel
Sonam Ninad Kulkarni
Georg Langebrake
Dr. Robert Rollins
Dr. Matthias Helfried Weissensteiner
Miriam started as Director of the IAR in 2020, and in parallel is hosting her Max Planck Research Group Behavioural Genomics at the MPI for Evolutionaray Biologie. The MPI based members of her team are listed here.